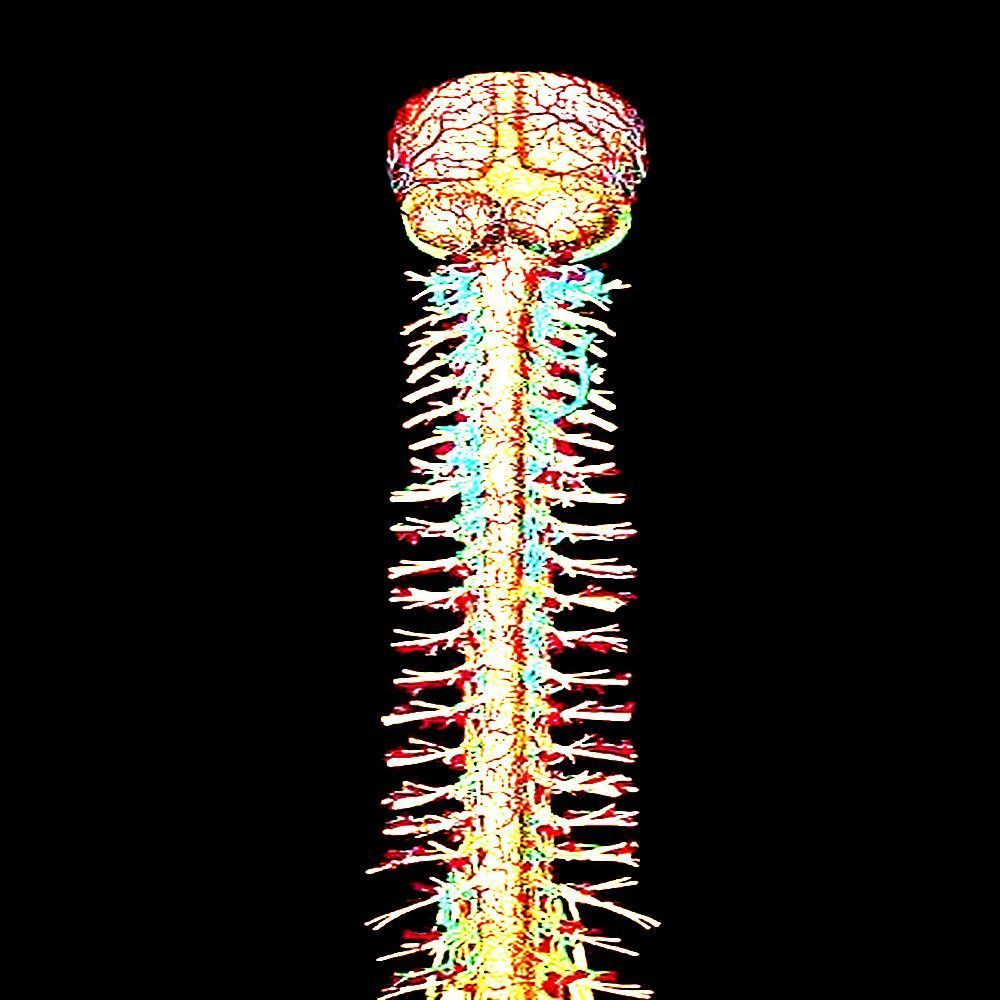
The subject is placed prone, neck hyperextended over a bolster, skin already prepped with iodine that stains like old bruising. The incision is not gentle — a 4cm vertical split down the posterior cervical midline, scalpel dragging through dermis, subcutaneous fat yellowed and glistening, until the trapezius and semispinalis cervicis muscles are reached and retracted apart with self-locking spreaders that crank open like a medieval instrument.
The laminae of C4 and C5 are exposed — pale ivory bone slick with periosteum. A surgical drill whines as it bores two anchor points, bone dust curling in the suction field. The CNB-7 housing, roughly the size of a thumbnail and milled from biocompatible titanium-graphene composite, is seated into the recess and locked with two ceramic micro-screws torqued to exactly 0.4 newton-meters.
The real work begins with the nerve roots. The dura is nicked with a micro-blade — a single precise slit, cerebrospinal fluid welling up clear and immediate, controlled with a temporary tamponade sponge. The C5 and C6 nerve roots are visualized: off-white, slightly translucent, thick as cooked spaghetti, branching from the spinal cord like rivers on a map.
Sixteen polymer-coated filament electrodes — each finer than a human hair — are threaded using magnetically-guided micro-forceps. They do not pierce the nerve so much as weave alongside the fascicles, the insulating myelin sheath intact and undisturbed. Each filament seats in a different functional cluster: motor efferents, sensory afferents, autonomic fibers running alongside. The device reads impedance continuously; a bad placement registers as a tone in the surgeon's earpiece.
Once all sixteen channels are confirmed, the filaments are bonded to the nerve roots using a photopolymer adhesive activated by UV pulse — three seconds of violet light and it cures rigid, locking the interface in place for the projected 40-year lifespan of the implant.
The housing is sealed. The muscle layers are closed in sequence with absorbable suture. The skin is stapled.
The patient is awake within six hours. The headache is described as having a railroad spike behind both eyes. By the third day, they can feel the device — not as pain, but as presence, a low electromagnetic hum behind thought, like a second heartbeat slightly out of phase.
Calibration takes another two weeks. The pilot interface doesn't simply read intention — it learns the specific electrochemical signature of this nervous system, this person's particular way of forming a decision before the muscles ever move. By the end of the calibration window, the lag between thought and machine response is under 11 milliseconds.
Phase I: Acute (Days 1–14)
The first sign something is wrong is usually phantom sensation. Not pain exactly — more like the ghost of a limb that never existed. Patients report feeling a third arm originating from the base of the skull, reaching forward into empty space. The brain, suddenly flooded with novel electrochemical input it has no vocabulary for, invents anatomy to explain the noise.
Electrode drift is the most common mechanical failure. A filament migrates 0.2mm — imperceptible on imaging — and suddenly the motor cortex is receiving sensory data on a channel it was never designed to interpret. The patient attempts to lift their right hand. Instead they smell copper. They try again. The copper smell intensifies until it becomes a taste, then a sound — a low metallic chord that they will later describe in intake interviews as "the color of something dying."
Phase II: Integration Failure (Weeks 2–8)
Approximately 12% of recipients develop what field technicians quietly call signal bleed: the implant's carrier frequency begins cross-talking with adjacent neural architecture. The result is the device's operational data — latency readouts, bandwidth logs, error codes — leaking into conscious experience as hallucination.
Recipients see loading bars in their peripheral vision that never complete. Error messages surface in dreams, rendered in no alphabet they recognize but understood intuitively, the way one understands dream-language. One documented case: a pilot who began experiencing his own heartbeat as a buffering icon. He could not distinguish between cardiac arrhythmia and packet loss. He stopped trusting his body to be real hardware.
Technologically-Induced Psychosis (TIP Syndrome)
The most catastrophic complication, occurring in roughly 4% of installations, has been given the clinical euphemism Integration Dissociation Disorder — though installers call it what it is: wire madness.
The mechanism is understood but not yet solvable. The CNB-7 operates on a feedback loop: it reads neural intent, executes it in the machine, receives the machine's response, and writes that response back into sensory cortex. In stable patients this loop is seamless. In TIP patients, the loop begins to echo. A thought triggers a machine response which writes back into the brain as a new thought which triggers a new machine response — recursive, accelerating, a signal bouncing between two mirrors facing each other.
The subjective experience, per survivor testimony:
- Voices that speak in pure functional logic. Not words. Instructions. Imperative statements delivered in the patient's own inner voice but felt as foreign, authored by something adjacent to the self. Execute. Confirm. Retry. Execute.
- Temporal fragmentation. The 11-millisecond machine-response lag, normally imperceptible, becomes enormous — a gulf between intention and action that the brain interprets as evidence that it is not the one doing the intending. Classic derealization: I watched my hand move. I did not move it.
- Sensory aliasing. Touch feels pixelated. Patients describe textures as having resolution — rough surfaces feel high-fidelity, smooth surfaces feel compressed, somehow lossy, as if their fingertips are reading an artifact of a thing rather than the thing itself.
- Identity partitioning. In the most advanced cases, patients report the gradual emergence of a second perspective — not a voice, but a viewpoint, sitting slightly behind and to the left of their own consciousness, watching. Silent. Evaluating. They do not believe it to be the device. They believe it to be what the device has made them into, looking back at what they were before.
By this stage, standard psychiatric intervention is functionally useless. The patient's reality-testing architecture — the very neural substrate used to distinguish internal from external, self from signal — is the compromised tissue.
Late Stage
Removal is rarely the clean solution laypeople assume. The photopolymer bond has integrated with the nerve root tissue by week six. Extraction means tearing, and tearing means deficit — motor, sensory, or both. Some patients choose to remain installed and simply adapt to a consciousness that now runs on two operating systems that were never meant to share memory.